Provider Resources


Our Invitation to Physicians and Researchers
We welcome physicians and researchers to refer their patients to the International Rare Brain Tumor Registry. Once your patient is enrolled, you’ll provide our team with their key biological, clinical and radiological data to be entered into the registry.
From the outset of a rare brain tumor diagnosis, the registry tracks information from the biorepository into an analysis of clinical data and more, ultimately leading to treatment innovations.
What is the criteria to participate?
To be eligible to participate in the registry, patients must be younger than 46 years old at the time of diagnosis and be diagnosed with one of the following rare conditions:
- CNS sarcoma
- BCOR-altered tumor
- Astroblastoma/MN-1 altered tumor
- Histologically ambiguous/unclassifiable tumor
- PLAG and PLAGL tumor types
- Other rare brain tumor
What does the registry collect?
Clinical data including:
- Demographics, tumor location, staging, pathology and molecular reports
- Imaging, such as MRI imaging
- Treatment information and disease course
Biospecimens (optional):
- Tumor tissue
- Blood
- Cerebrospinal fluid (CSF)
For patients enrolled in our prospective cohort, providers will also be asked to submit updates to the registry twice a year.
Participating Sites in the International Rare Brain Tumor Registry
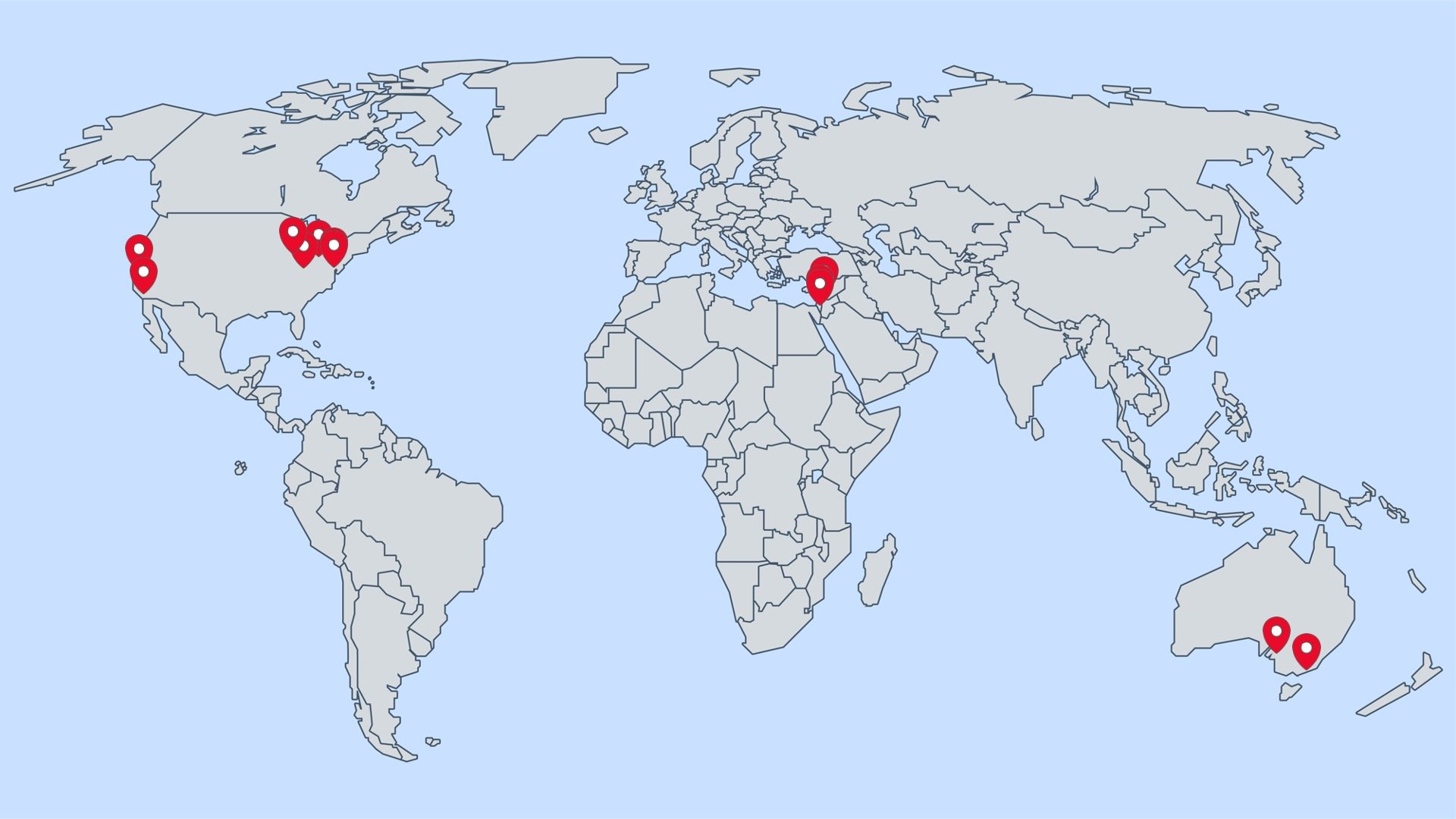
Participating Research Sites
Presentations and Publications
-
Intracranial mesenchymal tumor, FET::CREB fusion-positive: An integrative analysis of 81 cases Sharika Rajan, Hye-Jung Chung, Zhichao Wu, Omkar Singh, Karen Dazelle, Zied Abdullaev, Manoj Tyagi, Christina K. Ferrone, Mark Raffeld, Ina Lee, Jeffrey Gagan, Jie Chen, Sahara Cathcart, Caterina Giannini, Aivi Nguyen, Murat Gokden, Arie Perry, Igor Lima Fernandes, Angelica R. Putnam, Kyle Kurek, Richard M. Green, Charles Eberhart, Calixto-Hope Lucas, Ignacio Gonzalez-Gomez, Giselle Yvette Lopez, Karra Jones, Richard Prayson, Gabrielle Yeaney, Josephine Kam Tai Dermawan, Rati Chkheidze, Christina Appin, Erik J. Uhlmann, Alisa Taliansky, Melissa Blessing, Carrie Mohila, Jennifer Cotter, Jeremy Deisch, Felipe Andreiuolo, John R. Crawford, Christopher Mount, Anat O. Stemmer-Rachamimov, Nelli S. Lakis, Robert Schmidt, Geeta Chacko, Robert Newbury, Stewart Neill, Bryan Morales, Roger Fecher, Emily A. Sloan, David A. Solomon, MacLean Nasrallah, Martha Quezado, Adriana Fonseca, Kenneth Aldape. Neuro-Oncology, noag001, https://doi.org/10.1093/neuonc/noag001 (January 2026)
-
Molecular and clinical stratification of astroblastomas: Three distinct fusion-defined groups informing risk-adapted treatment strategies. Aniello Federico, Felix Schmitt-Hoffner, Adriana Fonseca, Neal Geisemeyer, Katharina Bruckner, Monika Mauermann, Martin Sill, Damian Stichel, Dominik Sturm, Ulrich Schüller, Arnault Tauziede-Espariat, Pascale Varlet, David Capper, Zied Abdullaev, Daniel Schrimpf, Florian Selt, Lane Williamson, Andrew M. Donson, Manila Antonelli, Evelina Miele, Matija Snuderl, Sebastian Brandner, Maria Łastowska, Jasper van der Lugt, Jens Bunt, Christof Kramm, Alexandra Kolenova, Aditya Raghunathan, Yelena Wilson, Lauren Weintraub, Jordan R. Hansford, Sabine Spiegl-Kreinecker, Barbara Aistleitner, Lorena Baron, Michal Zapotocky, Vijay Ramaswamy, Andrey Korshunov, Barbara Jones, Mimi Kjaersgaard, Mariëtte E. Kranendonk, Christine Haberler, Roger J. Packer, Natalie Jäger, Andreas von Deimling, Felix Sahm, Jan Koster, Kenneth Aldape, Stefan M. Pfister, Katja von Hoff, Johannes Gojo, Marcel Kool. Neuro-Oncology, noaf283, https://doi.org/10.1093/neuonc/noaf283 (December 2025)
-
Key Points
-
Astroblastoma, MN1-altered, comprises three molecularly and clinically distinct subgroups.
-
Gene fusions, including ones without MN1, define astroblastoma subgroups.
-
Different clinical outcomes and therapeutic effects suggest diverse therapeutic strategies.
- Clinical and Molecular Characterization of DICER-1 mutant CNS sarcomas, International Society of Pediatric Oncology, Philadelphia (June 29, 2024)
- Pediatric Neuro-Oncology Symposium, Washington, D.C. (May 2024)
- Clinical and molecular characterization of dicer-mutant central nervous system sarcoma. Dowiak* A, Williamson* L, Povilaikaite* J, Cabrera E, Aponte N, Garcia J, Quiroz L, Piña M, Martinez C, Valencia D, Barragan L, Reséndiz AB, Magelssen H, Trujillo Á, Osorio D, Figueredo O, Packer R, Sarmiento I, Suarez A, Mushtaq N, Abdullaev Z, Poria D, Bartels U, Bouffett E, Polo BR, Aldape K, Ramaswamy V, Fonseca A. ETMR-30.Neuro-Oncology doi: 10.1093/neuonc/noae064.200. PMCID: PMC11183751. (2024 Jun 18;26 (Suppl 4):0.)
Call for Collaborators
Interested in Becoming a Partner?
If you are a physician or researcher interested in partnering with the International Rare Brain Tumor Registry, please complete our form.




